Fat loss (lipoatrophy)
Fat loss is no longer reported with modern HIV treatment. Associated drugs: d4T (stavudine), AZT (zidovudine), possibly efavirenz.
Modern HIV drugs do not cause fat loss.
Symptoms
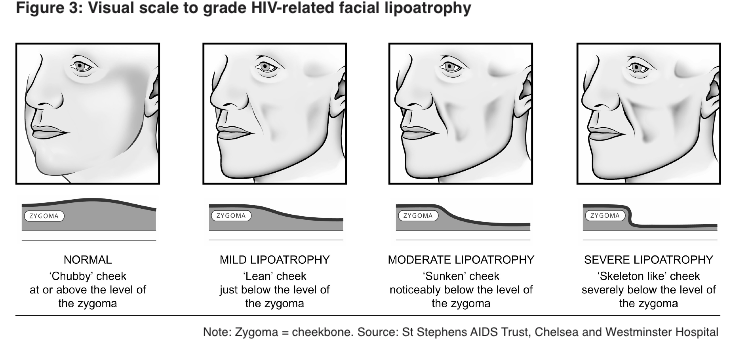
Lipoatrophy is the medical term for fat loss.
This is a side affect that was associated with older HIV drugs d4T and AZT.
Lipoatrophy is not linked to current treatment in the UK.
Symptoms included loss of fat from under the skin on your arms and legs, which can make your veins look more prominent. It also includes loss from the face, especially sunken cheeks and temples.
Fat can be lost from the soles of the feet making walking more painful and tiring.
Role of d4T and AZT
Clinical lipoatrophy – where you can see a change in body fat – is common after using either d4T or AZT for more than six months. Both drugs change the way that fat cells are produced and develop.
At a cellular level this can occur after only a few weeks or months of treatment.
Nucleosides (nukes) have been shown to damage the energy producing part of healthy cells called mitochondria.
In most studies, d4T damages fat cells at around twice the rate compared to AZT. d4T might also lead to lipoatrophy that is more difficult to reverse because it might damage cells at an earlier stage of their development.
Other nukes?
Not all nukes cause lipoatrophy. This is not a side effect of 3TC, FTC, tenofovir and abacavir. The role of ddI is unclear.
The risk of lipoatrophy for people who are starting their first treatment should now be very low in Western countries.
Newer drugs do not cause this side effect, and increased monitoring should pick this up if you are using older drugs like AZT.
Neither d4T or AZT are recommended as routine first-line therapy in the UK, unless specific health complications require it. People currently using either of these drugs should be offered alternatives.
Other HIV drugs and fat loss
Some studies reported a higher risk of fat loss when d4T or AZT were used with protease inhibitors.
The US study ACTG 5142 reported higher rates of fat loss in people using efavirenz compared to lopinavir/r, even when use of nucleosides were taken into account. These findings are not fully understood.
Several studies have reported higher rates of lipodystrophy in people using combinations that include three drug classes–nukes, NNRTIs and PIs.
Switching HIV treatment
Switching d4T or AZT to either abacavir or tenofovir, or using other combinations of drugs, can reverse the fat lost in limbs.
Reversing fat loss from the face or buttocks is more difficult, but this might be possible if you switch treatment early.
Switching is very safe, but the choice of new drugs needs to consider your previous treatment history to minimise the risk of drug resistance.
Any reversal of the fat loss is likely to take at least six months to become noticeable. This is because these symptoms developed slowly and if they are going to reverse this will also take time.
In studies where people switch, approximately +0.3 kg can be detected by scans at 6 months. In one study it took about two years (with an increase of +1.3 kg) before these patients noticed a difference.
Injectable treatments
Many of these are used without approval for treating HIV-related fat loss.
Many substances have been used to treat HIV-related fat loss in the face but very few have been carefully researched.
Although non-permanent products need top-up treatment, these are currently the safest option. They work with your natural ageing process. Unlike permanent implants, there is no risk of it moving.
- In the US, only New-Fill and Radiesse have been approved to treat HIV-related facial lipoatrophy.
- In the UK, New-Fill is the most widely used, and it is approved by some NHS trusts, we focus on this product in this guide. It is also supported by the strongest safety and efficacy results.
New-fill (Sculptra)
New-fill (polylactic acid, PLA) has shown promising results in correcting the effect of facial fat loss and is approved in the US as a treatment for HIV-related lipoatrophy. Most people require 4-5 sets of injections but severe cases might require more sessions.
New-fill does not replace fat but generates new collagen growth. This gives the effect that your skin grows thicker, sometimes by up to 1cm. This process continues for several months after the injections have finished.
New-fill has also been used to correct fat lost on the soles of the feet.
New-fill is available free on the NHS in many of the larger HIV clinics in the UK. These include Brighton, Manchester, and any patient attending a London clinic. Since 2005, New-fill has been available free on the NHS for any patient registered at a London clinic.
UK HIV treatment guidelines recommend that corrective treatment or surgery should be provided on the NHS.
However, New-fill is not equally available throughout the UK. You might have to lobby you doctor and NHS trust. You might decide to register at a new HIV clinic to access this treatment.
Private treatment costs vary by clinic. Private treatment should ONLY be from a practitioner with experience treating HIV-related lipoatrophy.
Other injectible compounds
Radiesse
A second non-permanent filler approved in the US to treat HIV-related facial fat loss is called Radiesse. This is the trade name for a formulation of calcium hydroxylapatite suspended in a gel.
Although this is used in some private clinics in the UK, it is not approved by the London commissioners as a free NHS treatment.
Bio-Alcamid
Bio-Alcamid (polyalkylimide, Polymekon) is a ‘gore-tex’ filler that was used briefly but has now been linked to serious complications in 10% people. These relate to infections in the implant, often years after the procedure.
BioAlcamid no longer being used or recommended in many countries including the UK. See: https://i-base.info/htb/14135
Although the manufacturer claims that BioAlcamid can be removed, it is a permanent implant because removal is traumatic and becomes increasingly difficult over time.
Anyone who has used BioAlcamid should inform their dentist about their implants and not have dental injections close to the implant site.
Other complications have been reported from trauma. Do not take up boxing or contact sports.
BioAlcamid has probably been used by several hundred people in the UK, and several thousand people in Europe and the US. Information is difficult to assess because this was largely in private clinics.
Silicone
Other approaches try to inject or implant material (fat or silicone) and hope it will stay in position. Very often, it disperses, moves or appears lumpy.
Silicone injections are dangerous and ineffective and were banned in the US many years ago.
A fine grade formulation called Silikon 1000 Microdroplets was studied in the US but further results were not available when updating this guide.
Fat transfer (Coleman technique)
Fat transfer involves extracting fat from one body site and reinjecting it surgically in another. This is usually subcutaneous fat from the stomach, which is then transplanted to the face.
Fat that has accumulated as a result of lipodystophy is not suitable for transplanting.
Although the results were very good the process is now less frequently used. This is because it is invasive and more expensive.
Further info
A US community site with information on lipoatrophy treatment:
Last updated: 1 March 2023.