Cases of mpox reinfection in HIV negative men on PrEP: atovaquone, mefloquine and molnupiravir as potential treatments
6 April 2023. Related: mpox.
Simon Collins, HIV i-Base
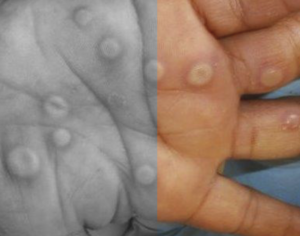 A case of mpox reinfection in an HIV negative gay man without immunosuppression, has just been reported in CID, occurring six months after the first infection. [1]
A case of mpox reinfection in an HIV negative gay man without immunosuppression, has just been reported in CID, occurring six months after the first infection. [1]
A similar case was also reported in the BMJ earlier this year, with only four months between infections, during which time the man had also received two doses of the Jynneos vaccine. [2]
The first case was a 31-year-old man on PrEP living in Switzerland who was first diagnosed with mpox in May 2022, at the outset of the epidemic last year. Presenting symptoms included four umbilicated penile lesions. Skin lesions and pharyngeal swabs tested PCR-positive for mpox with a cycle threshold (Ct) of 16.5 and 35.3 respectively. He also tested positive for chlamydia. Within two weeks skin lesions spontaneously resolved without complications.
On 1 December, he was diagnosed with mpox a second time and proctitis after reporting anal pain without bleeding that starting two weeks earlier, four weeks after a visit to Brazil – where a very high number of daily infections were still being reported. Rectal swabs tested PCR-positive for mpox with a Ct of 27. He also tested positive for chlamydia again. Four weeks after exposure he had an anal fissure but no typical mpox lesions.
On 13 December, orthopoxvirus PCR was negative in the blood and urine.
Unfortunately, viral isolates were only available for the first infection. The study authors discussed the potential that the second infection was due to immune escape mutations or that the high viraemia associated with anal lesions might have outweighed any earlier immune response. However, the median time for mpox PRC-positivity in rectal swabs is about 8 days with a 95th upper percentile of 14 days and no cases reporting persistent viral shedding over months.
The second infection was described as mild and resolved completely.
Another case of mpox reinfection in an HIV negative man on PrEP was reported in the BMJ earlier this year.
In this case, there was only four months between the two infections, during which time this person had also received two doses of the Jynneos vaccine.
One dose was given a week after the first infection in July 2022 and the second was given 10 weeks later in October (5 weeks before the second infection and 9 weeks before painful anal lesions).
Both mpox infections resolved without complications.
comment
These cases show that the hoped-for sterilising immunity after an initial systemic infection is not sufficiently protective to prevent subsequent infections, even after less than six months.
Even though this is only a single case, the lack of protection more than four weeks after the second vaccine shot is also worrying. Although several reports claim high efficacy for the vaccine, protection has not been properly tested as mpox cases were already falling before significant protections would have been established.
Early studies reporting high levels of efficacy are also observational datasets, where the people receiving vaccinations were at lower risk of mpox compared to the unvaccinated groups where higher mpox rates were reported. [3, 4, 5]
This was supported by numerous studies at CROI 2023 reporting that people at highest need for mpox vaccinations in the US were least likely to be able to access them. [6, 7, 8, 9, 10, 11]
It was also interesting to see a recent CID paper suggesting that atovaquone, mefloquine, and molnupiravir might have activity against mpox and with greater potency than cidofovir. [12]
As this issue of HTB went to press, two further cases of mpox reinfection were publihed in Lancet Infectious Diseases. [13]
A recent cluster of 17 cases of mpox in the Centre-Val del Loire region in France included 5/17 who had received two shots of the mpox vaccine in 2022. Another 5/17 cases had received a single vaccine dose and but also had a childhood smallpox vaccination. [14]
Another cluster reported from Chicago included that 9/13 mpox cases were in men who had received two vaccine doses. [15]
Reference
- Musumeci S er al. A case of mpox reinfection. Clinical Infectious Diseases 2023 ciad147. DOI: 10.1093/cid/ciad147.
https://pubmed.ncbi.nlm.nih.gov/36905148/ - Golden J et al. Case of apparent mpox reinfection. Sex Transm Infect. 2023 Jan 27:sextrans-2022-055736. doi: 10.1136/sextrans-2022- 055736. (27 January 2023).
https://sti.bmj.com/content/early/2023/01/26/sextrans-2022-055736 - Payne AB et al. Incidence of monkeypox among unvaccinated persons compared with persons receiving ≥1 JYNNEOS vaccine dose—32 U.S. jurisdictions, July 31–September 3, 2022. MMWR Morb Mortal Wkly Rep 2022;71:1278–82. https://doi.org/10.15585/mmwr.mm7140e3PMID:36201401
- Payne AB et al. Reduced Risk for Mpox After Receipt of 1 or 2 Doses of JYNNEOS Vaccine Compared with Risk Among Unvaccinated Persons — 43 U.S. Jurisdictions, July 31–October 1, 2022. MMWR Morb Mortal Wkly Rep 2022;71:1560–1564. DOI: http://dx.doi.org/10.15585/mmwr.mm7149a5
- Arbel R et al. Effectiveness of a single-dose modified vaccinia Ankara in human monkeypox: an observational study. Research Square [Preprint posted online September 23, 2022]. NOTE – this preprint is still unpublished after six months.
https://www.researchsquare.com/article/rs-1976861/v2 - Vaidya A et al. Characteristics and disparities among hospitalized persons with mpox in California. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 902.
https://www.croiconference.org/abstract/characteristics-and-disparities-among-hospitalized-persons-with-mpox-in-california
- Philpott DCE et al. CD4 count < 350 cells/mm3 increases risk of hospitalization with mpox in PWH. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 903.
https://www.croiconference.org/abstract/cd4-count-350-cells-mm3-increases-risk-of-hospitalization-with-mpox-in-pwh - Corma-Gómez A et al. Mpox virus infection is more severe in patients with uncontrolled HIV infection. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 904.
https://www.croiconference.org/abstract/mpox-virus-infection-is-more-severe-in-patients-with-uncontrolled-hiv-infection - Silva MST et al. Impact of HIV infection on mpox-related hospitalizations in Brazil. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 905.
https://www.croiconference.org/abstract/impact-of-hiv-infection-on-mpox-related-hospitalizations-in-brazil - Garneau WM et al. Clinical outcomes among in- and outpatients with mpox in an urban health system. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 907.
https://www.croiconference.org/abstract/clinical-outcomes-among-in-and-outpatients-with-mpox-in-an-urban-health-system - Cholli PA et al. Characteristics of patients hospitalized with mpox during the 2022 us outbreak. CROI 2023, 19–22 February 2023, Seattle. Poster abstract 912.
https://www.croiconference.org/abstract/characteristics-of-patients-hospitalized-with-mpox-during-the-2022-us-outbreak - Akazawa D et al. Potential anti-mpox virus activity of atovaquone, mefloquine, and molnupiravir, and their potential use as treatments, Journal of Infectious Diseases, 2023; jiad058.
https://doi.org/10.1093/infdis/jiad058 -
- WHO. Multi-country outbreak of mpox.Situation report. (13 April 2023).
PDF link. - US CDC. Potential risk for new mpox cases. (16 May 2023).
https://www.hiv.gov/blog/potential-risk-for-new-mpox-cases
This report was first published on 15 March 2023. It was updated on 11 April and 12 May to include the paper in Lancet Infectious Diseases and the clusters reported in France and Chicago.

