Tables and diagrams
We’ve collected key tables and diagrams in the guide here for ease of reference.
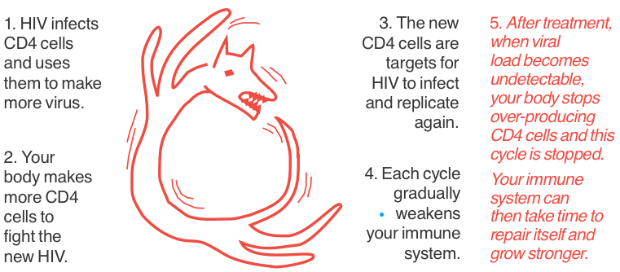
When not on treatment, your immune system works in overdrive
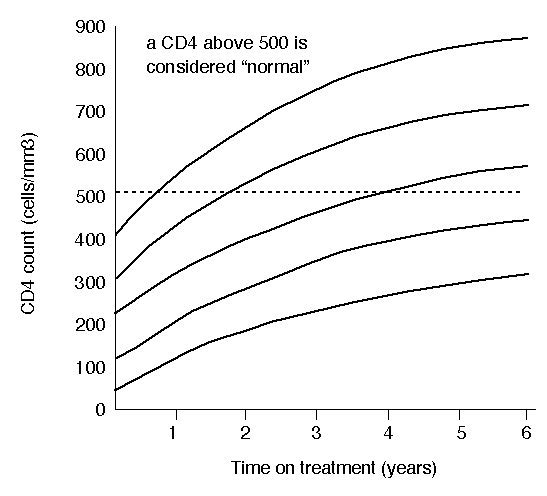
CD4 increases on ART
Drug levels with good adherence
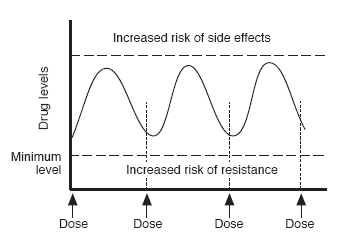
Drug doses are calculated so that average drug levels are high enough to be active against HIV for 24 hours a day. They are also low enough to minimise the risk of side effects.
A missed or late dose increases the risk of resistance
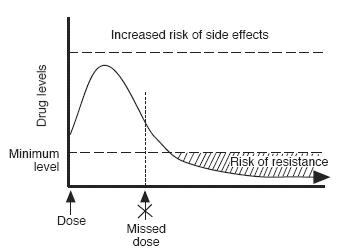
Missing or being late with a drug lets the drug levels fall to a level where resistance can develop. The more often you are late, the greater the chance of resistance.
The main types of hiv drugs
| Abbreviation | Full name(s) |
|---|---|
| NRTIs/NtRTIs (“nukes”) | nucleoside/tide reverse transcriptase inhibitors or nucleoside/tide analogues |
| NNRTIs (“non-nukes”) |
non-nucleoside reverse transcriptase inhibitors |
| PIs | protease inhibitors |
| INIs (or INSTIs) | integrase strand transfer inhibitors |
| CCR5 inhibitors | CCR5 inhibitors are a type of entry inhibitor |
| mAbs | monoclonal antibodies – which also block entry to the CD4 cell |
| Fusion inhibitors | fusion inhibitors are a type of entry inhibitor |
| PK boosters | Ritonavir or cobicistat are included in some combinations to boost another drug |
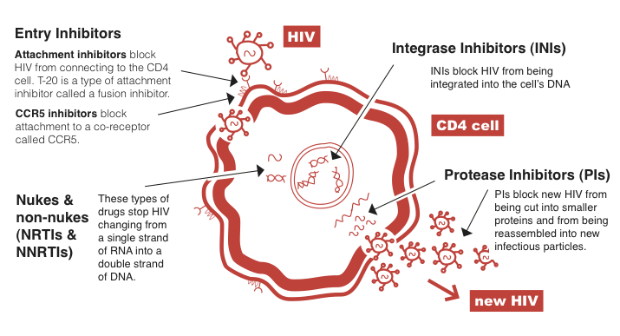
HIV lifecycle – how drugs work in different ways
If a CD4 cell is infected by HIV, this cell is used to produce hundreds of new copies of HIV. Different drugs block different parts of this HIV life cycle.
Last updated: 1 November 2024.
