AIDS 2012: non-technical conference summaries: HTB supplement
1 August 2012. Related: Special reports.
 Download PDF file (1.1 MB)
Download PDF file (1.1 MB)
This supplement is a non-technical review of some of the most interesting studies presented at this meeting.
Technical reports from the conference are published in HIV Treatment Bulletin, available online and to order in print from i-Base.
https://i-base.info/htb/keyword/world-aids-19-washington-2012
Hyperlinks in this PDF file link directly to each abstract or session.All abstracts and webcasts can be seen on the conference website.
 Introduction to AIDS 2012
Introduction to AIDS 2012
This supplement covers some of the presentations, studies and events from the 19th World AIDS Conference.
Held every two years and organised by the International AIDS Society (IAS) this is the largest HIV medical meeting.
Links for further information and to the online coverage are included throughout.
Although originally an exclusively medical meeting, over the last decade, the programme has increasingly broadened to include social and political aspects of HIV.
Approximately 85% of over 3500 studies are now related to human rights, funding, policy, prevention, access to treatment and issues of stigma.
The majority of treatment-related studies were posters (a summary presented on a 2 x 1 metre display). Only 25 posters each day focused on basic science (Track A) and less than 75 on clinical studies (Track B). From over 80 hours of podcasts only five sessions are on treatment – perhaps showing how effective treatment already is.
So although we report important scientific research, the webcasts from the social, political and human rights sessions provide the context for the main meeting.
With more than 22,000 delegates and thousands of studies and presentations, navigating the conference was sometimes frustrating. But within a few hours of the closing sessions the halls were empty and the venue prepared for computer games booked for the following week (where IAS stands for Increased Attack Speed). And it became easier to distill the point of all this diverse activity.
With World AIDS conferences, certain issues usually come to represent the meeting rather than headline results based on new scientific advances.
This year included more people talking about ending AIDS (at least since 1998 when I first attended). But 30 years into the epidemic the conference theme is only tentatively aiming at ‘Turning the Tide’. The optimism is still early days. Even with the tremendous advances in treatment and prevention and exciting early research into a cure, HIV will be with us for a while. Unfortunately, the AIDS-free generation that was referenced in many presentations, still looks unlikely to become a reality anytime soon.
However, treatment that has now been shown to both save lives and reduce infections. Expanding access to treatment makes one conference focus this year as the ability to mark 2012 for achieving 8,000,000 people on treatment.
Remembering the impossibly slow progress of the “3×5” campaign (to get 3 million people on treatment by 2005), it is significant that this conference, with its shift to focus on treatment access has sailed past this once-doubted goal.
Programme strengths this year included:
A platform for speeches
On policy and access, and for HIV positive people and activists from many different backgrounds and circumstances, leading many of the community responses to give their diverse perspectives on a world stage.
The best are passionate and rational strategies for change.
Asserting the focus on a cure
Many sessions included early research related to a cure. This included a pre-meeting workshop and a scientific plan for the research goals and objective to this ultimate challenge.
Clinical data
Highlights included new drugs for HIV and TB, children’s health and other studies.
HIV prevention
With an emphasis this year focusing on policy and implementation rather than new clinical data. This especially focused on Treatment as Prevention (TasP), Pre-exposure Prophylaxis (PrEP), circumcision, needle-exchange, and infant and maternal access to treatment.
HIV and long-term health
The increasing focus on inflammation as a concern from untreated HIV and long-term health. This overlaps with ageing and the earlier use of HIV treatment.
To launch publications and reports
Many publications contain more detail and planning that could fit into a single symposium or poster. All are free online.
Other community events
The conference incuded a “Global Village” for many community events. This included performances and screenings.
This year as part of the conference, more than 50,000 quilts hung in the conference halls and were laid out along part of the National Mall Park near the Washington Memorial and 50 other locations in the city.
19th World AIDS Conference, Washington DC
Human rights, access to treatment, activism and politics

If you can be moved by carefully chosen words, focused on strategies for change, that elevate human rights with the dignity they deserve, then take time to watch some of the webcasts from the meeting.
This conference has developed the highest profile for public speeches on policy and most sessions are online.
More than 80 sessions are available online. Most vary from 1-2 hours for sessions and from 5 to 30 minutes for individual speeches. When available, IAS webcast links are to the individual talk (rather than to the whole session which occurs for Kaiser Foundation webcasts).
Here are links to a few highlights.
Opening programme
http://pag.aids2012.org/session.aspx?s=53
The auditorium was filled for this diverse and moving programme that included the Gay Men’s Choir (Washington) and 24 year old Annah Sango from Zimbabwe talking about her hopes as an HIV positive woman.
Also, Jim Yong Kim, current president of the World Bank (and the first to address the AIDS conference) and former director of HIV/AIDS department at the WHO, talks about why an end to AIDS should be more a reality now than “3×5” was in 2002, and how the World Bank has a commitment to ending AIDS and poverty.
Françoise Barré-Sinoussi (IAS)
http://pag.aids2012.org/flash.aspx?pid=368
When speaking about her 2008 Nobel laureate for medicine for her role in discovering HIV, this virologist, advocate and IAS president for the next year, said “it does not belong to me but to all of us”. (Difficult to imagine this from Robert Gallo).Outlining priorities for the next year, she made it clear that we already have the scientific understanding to end AIDS:
“As a scientist, in 2012, it is unacceptable that more than 300,000 babies are born HIV infected each year when we have had, since the 1990s, the tools to prevent mother to child transmission; that risk reduction strategies including needle exchange programmes are not implemented everywhere when we know this is one of the most effective scientific interventions to prevent HIV infection in IV drug users; and that intellectual property rights undermine access to high quality medicines and diagnostic tools in resource limited settings”.
Bill Clinton
Plain-speaking assertion that continued treatment roll-out is both achievable and affordable from one of the key movers behind price reductions for first and second-line ART.
http://pag.aids2012.org/flash.aspx?pid=365
Phill Wilson
HIV positive US activist, highlighting the complexity of HIV healthcare in the US where young gay men, especially if they are black, continue to have some of the highest rates of new infections and that their lives can still be cut short: 60% of black MSM in the US are likely to be HIV positive by age 40.
http://pag.aids2012.org/flash.aspx?pid=1558
Hilary Clinton
[Talking over a protest against US trans-pacific trade deals that limit access to generic drugs]: “What would an AIDS conference be without a little protesting? Part of the reason we have come as far as we have is because so many people all over the world have not been satisfied that we have done enough, and I am here to set a goal for a generation that is free of AIDS.”
An essential talk to understand the changing US political approach to HIV, notably distinct from an “emergency” response (the “E” in PEPFAR). “If we want to save more lives we have to go where the virus is […] And that means science should guide our efforts”. Both political and personal. (78 minutes into the Turning the Tide session on 23 July).
http://globalhealth.kff.org/AIDS2012/July-23/ending-the-epidemic.aspx
Rolake Odetoyinbo
“Women with HIV have come a long way, from being zero – lower than trash to having seats at the table – we had to earn our place […] We have fought for this but also salute those people who have helped open doors… We did not ask to be the face of HIV, but 60% of HIV positive people in Africa are women. Women are 80% of care providers – doctors, nurses and counsellors – grandmothers and mothers and sisters and people leaving school to care for our dying relatives.”
In a session of on women in leadership that included a video message of support from Aung San Suu Kyi and an address by Laura Bush, Rolake, 50 minutes in, is the one not to miss.
http://globalhealth.kff.org/AIDS2012/July-26/leadership.aspx
New drugs and formulations
At every conference we want to know about new drugs and how they might improve future care. In Washington we learnt about new integrase inhibitors and a new booster that could be an alternative to ritonavir.
• New booster – cobicistat as an alternative to ritonavir
Most protease inhibitors (PIs) need to be boosted to reach effective levels. Up until recently the only options to do this was to use low doses of another PI called ritonavir.
A new option close to approval is called cobicistat (pronounced COB-IZ-EE-STAT). Cobicistat was compared to ritonavir to boosting atazanavir in almost 700 people stating their first treatment. Everyone also used tenofovir/FTC in the combinations.
Although both boosters look similar in terms of efficacy and side effects, there is a practical advantage for the new drug. The company producing cobicistat is working with the manufacturers of darunavir and atazanavir to coformulate the PI and booster in the same pill.
This may help with fewer pills, fewer prescriptions and a slightly easier life for people taking these PIs.
• New integrase inhibitors
Two new integrase inhibitors that are also likely to be close to approval had new studies presented at the conference. Both are once-daily meds.
Elvitegravir is a new integrase inhibitor that is included in the 4-in-1 single tablet combination called Quad. A study in Washington reported that after two years elvitegravir was similar to raltegravir in over 700 HIV positive people with drug resistance.
Elvitegravir has already been submitted to the FDA for approval as a separate drug, and shortly after the conference Quad was approved in the US (tradename Stribild). The decision on European approval is expected by the end of 2012.
Dolutegravir is an integrase inhibitor that is being developed in a single tablet combination with abacavir and 3TC. In Washington the Spring-2 study reported dolutegravir to be similar to raltegravir in people using treatment for the first time. About 90% of people using either drug had undetectable viral load after a year.
Importantly, both raltegravir and dolutegravir have formulations for use by children that were also presented at the conference.
d4T without the side effects: early days for a new nuke
Finally, several studies were presented for a compound that is similar in structure to d4T.
Although these are early days this is being developed because it is unlikely to have the same side effects associated with d4T, including neuropathy and fat loss.
Other treatment studies
Several studies presented new information on other aspects of HIV treatment.
• Maraviroc plus atazanavir/ritonavir
The CCR5 inhibitor maraviroc was initially studied in combination with two other active drugs. However, because some boosted protease inhibitors increase the drug levels of maraviroc so that only half the recommended dose is needed, some researchers used this as the basis for a new combination.
Unfortunately, the percentage of people with undetectable viral load looked worse compared to a three-drug combination (68% vs 82% with maraviroc plus tenofovir/FTC). New studies are now using darunavir/ritonavir with maraviroc to see if this can be improved.
• Gallstones with atazanavir
A French study reported that atazanavir was associated with development of gallstones.
Although this is the first report, the link seemed fairly convincing as analysis of the stones showed that they largely contained atazanavir. However many of these cases generally had other pre-existing liver complications, including hepatitis C.
Atazanavir has also been reported to cause kidney stones.
• Raltegravir good at five years
Several studies reported longer-term (five year) results using raltegravir. Of note, after four years raltegravir became significantly better than efavirenz (both also used with tenofovir/FTC). This was largely driven by more people stopping efavirenz due to side effects.
A study at a workshop prior to the main conference also reported that markers of muscle toxicity were higher in people using raltegravir (or reporting recent exercise) compared to people not using raltegravir.
The changes were all generally mild and no cases of serious muscle toxicity were reported. However, a more serious form of muscle toxicity called rhabdomyolysis has been reported with raltegravir, so this might be an early signal to watch.
• Why does male circumcision protect from HIV?
One study proposed a new mechanism for why medical male circumcision reduces the risk for a man to catch HIV.
Previously this was thought to be due to a thinner keratin layer (of sort of tissue padding) in the inner foreskin.
The new study suggested that the route for infection is through the sensitive membrane of the head of the penis. Uncircumcised men were found to have a higher percentage of cells nearer to the skin surface, that HIV needs to find for infection to take hold.
 Publications launched at IAS
Publications launched at IAS
The IAS conference is used to launch publications on important aspects of HIV treatment or access. Here are a few that caught our attention. All are available free online.
Towards an HIV Cure: Global Scientific
Strategy
Officially announced at a pre-conference workshop, the IAS “Towards an HIV Cure” Global Strategy represents a thorough and comprehensive framework for cure-related research in the future.
 The report identifies seven main research priorities in how best to overcome HIV persistence before a cure (functional or sterilising) can be realised.
The report identifies seven main research priorities in how best to overcome HIV persistence before a cure (functional or sterilising) can be realised.
- Why does HIV persist: immune and viral factors
- Where does HIV persist: tissue and cell reservoirs
- Immune activation and dysfunction on ART
- Natural models of HIV/SIV control
- How to measure persistent infection
- How to reverse latency: treatments
- Immune approaches, gene therapy, vaccine
At the report launch, Steven Deeks, co-chair of the IAS cure working group, said “We are entering a stage in the epidemic in which we can seriously begin testing drugs that either prevent latency or which force the virus out of its hiding place, making it susceptible to our current drugs.”
http://www.iasociety.org/Web/WebContent/File/HIV_Cure_Full_recommendations_July_2012.pdf (PDF)
 MSF reports
MSF reports
The international health organisation Médecins Sans Frontières (Doctors Without Borders) launched several important documents on improving access to treatment. These included:
- The importance of viral load tests globally
- Untangling the web of ARV price reductions
- How US trade and intellectual property agreements further threaten global health
- An MSF report for faster scale-up
Lancet series on HIV in MSM
The Lancet published a series of articles highlighting the ongoing growth of the HIV epidemic in men who have sex with men (MSM).
The articles cover both the science and culture behind the MSM epidemic. One comment piece examines “The irony of homophobia in Africa”, which is reflected in a review of the recently released film “Call me Kuchu” which looks at the life and death of David Kato in Uganda.
Topics covered also include the unique challenges faced by black MSM around the world; stigma and discrimination affecting MSM; and a look at the global epidemiology of HIV infection in MSM.
http://www.thelancet.com/series/hiv-in-men-who-have-sex-with-men
 TAG/i-Base Pipeline report 2012
TAG/i-Base Pipeline report 2012
A plug for our own publication. For the third year i-Base have partnered with the New York-based activist group Treatment Action Group (TAG) to produce a combined pipeline review of new drugs, formulations, vaccines, strategies and diagnostics for HIV, hepatitis and TB.
http://www.i-base.info/2012-pipeline-report
UNAIDS report: Together we will end AIDS
Launched just prior to the conference, this report from UNAIDS was stacked high in the UNAIDS booth. It includes photographs, graphics and contributions from people with key roles in the global response.
The most useful information is probably the individual country data relating to new infections in children, the incidence in women, details on access to treatment and care, and HIV-related mortality. All on pages 122-123, just before references.
http://www.unaids.org/en/resources/campaigns/togetherwewillendaids
JAIDS supplement: HIV and ageing
A review of current knowledge and research concerns relating to HIV and ageing was published as a supplement in the Journal of AIDS.
Rather than the benefits of HAART that have extended life expectancy by 50 years, the authors focus on the complex interplay of social and medical circumstances whereby “on average, a 20 year old initiating ART may already have lost one-third of their expected remaining years of life compared to similar HIV uninfected persons”.
The report highlights the concerns about Inflammation, co-infection (including CMV), reviews current data on biomarkers (IL-6, CRP, d-dimer, soluble CD14 and markers of T-cell activation and senescence), highlights current knowledge gaps and suggests priority areas of future research.
http://journals.lww.com/jaids/toc/2012/07011
ACT-UP at the movies: ‘How to Survive a Plague’ and ‘United in Anger’

Poster still from How To Survive a Plague
Two new films on ACT-UP and early HIV activism in the US were screened during the week.
On the first evening, a few blocks south of the conference centre, a preview screening took place for How To Survive a Plague: a film that tracks the history of ACT-UP from the early meetings to the arrival of the first effective treatments.
The film tells the story of how people came together to organise to overturn the way that research, drug regulation, medicine and science worked in the US, fighting ignorance, indifference and prejudice. It is that simple and dramatic. And many activists who followed, myself included, recognise that little of the current work we are involved in today would have been possible without their achievements.

Early ACT-UP members attending the screening of How To Survive a Plague: (l-r) Sam Avrett, David Barr, Moisés Agosto, Mark Harrington, Garance Franke-Ruta and Gregg Bordowitz.
The director David France tracked down more than 700 hours of archived footage from 30 different collections, and edited this with recent interviews from some of early activists today. But many others in the film, just as crucial in the struggle, did not make it. As you are drawn into their story you meet people who then leave you to finish the journey without them.
ACT-UP came from a crisis that is difficult for anyone who was not there at the time to imagine now – but it did not come with the first cases, or even in the first years. By the time the group was founded in New York in 1987, six years after the first deaths from AIDS, 40,000 people had died in the US and 500,000 people had died globally.
But the same month that the group was formed, a previously existing compound called AZT that was developed with considerable public funding, was approved as the first treatment. But Boroughs-Wellcome set the price higher than any other current medication at $10,000 a year, and an ACT-UP demonstration closed trading on the floor of the New York stock exchange, forcing the price to be dropped by 20%.
Other actions used media attention to force political change. They unrolled a giant replica condom over the home of the homophobic Republican senator Jesse Helms, closed the FDA with thousands of demonstrators to change in the mechanism for regulatory approval for life-threatening conditions, held a “die-in” in St Patricks Cathedral in response to Catholic directives against using condoms, and in 1996, scattered the ashes of friends and lovers on the White House lawn as a last act of protest.
But the change came not just from shouting and protests – though this threat brought muscle to the groups demands. Behind the scenes, ACT-UP members became sufficiently expert at research and science, to be able to draft the first strategic agenda for HIV research and design the first commercial drug trials.
ACT-UP got the FDA to overturn its decision to refuse approval for a drug called DHPG (proven to save the eyesight of people with CMV, later called ganciclovir). And they forced open the formerly closed FDA committees – “why should science be shrouded in secrecy?” and set public involvement as a new standard. Even when scientists recognised that nothing was certain the interactions with activists led to personal responses that “we have no right to say we can’t do this”.
These examples show how ACT-UP’s mix of public protest and forcing dialogues with health officials, scientists and drug companies lead to faster, more effective, more affordable treatment. People organised together to say when things were not good enough.
In a panel discussion after the screening, director David France described how reactions to the film vary: “for those under 40 everything is new and for those older they are watching home movies”. He made the film “to correct a misdirection that not everything was about loss. It was a period of immense creativity, activism and change”. For all the noise and press, these activists were also quiet, considered, tactical, strategic.
A few days later “United in Anger: A history of ACT-UP” was screened in the Global Village at the IAS conference.
Produced by Jim Hubbard and Sarah Schulman of the ACT-UP oral history project and based on the extensive interviews with over 140 activists this is more of a formal history of the group. It includes more HIV positive women and other campaigns including the four-year drive to change the US CDC definitions for AIDS-defining illnesses to include infections that specifically affected women, without which they were excluded from public assistance programmes.
Both films are important to understand and appreciate our history. See them to be inspired that change can happen and for the importance of telling your own histories afterwards.
Edited from a longer review on the i-Base website.
https://i-base.info/simoncollins/2012/07/film-review-how-to-survive-a-plague
Four ways to find a cure
While ARVs extend life expectancy by 30 years or more, why is this not the same as for HIV negative people, and will there be a cure?
The recent increased focus on these questions, especially in relation to a cure, covers four main areas of research.
Some research looks at how to eradicate HIV completely but some is looking at a functional cure (where you would still be HIV positive but treatment could be stopped).
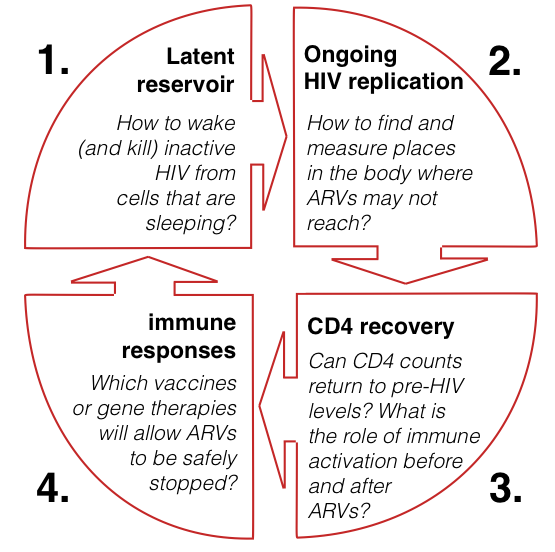
Within a few weeks of infection, HIV gets inside very long-lived CD4 cells that then enter a resting or sleeping stage that can be anywhere from dozing to in a coma. These cells are not affected by ARVs which is why treatment is lifelong.
Even one infected cell would be enough to restart infection, even if it was the only infected cell after 50 years of treatment.
One aspect of cure research is looking at how to wake the HIV in these cells and studies using vorinostat have shown this might be possible. Another looks whether HIV might still be reproducing in other parts of the body such as the gut.
Thirdly, researchers are trying to find out why CD4 counts do not completely recover after starting treatment. They are also looking at how HIV can cause the immune system to be over-activated when not on treatment, and whether this is reversed when viral load becomes undetectable. This research overlaps with many of the questions about HIV and ageing.
Finally, even if latent HIV is activated, a new immune response may still be needed. Gene therapy can make some CD4 cells resistant to HIV. Or a vaccine? A few people can control HIV without treatment either from infection or after stopping treatment and their responses may be key.
Cure research at the meeting was reported in HTB at this link:
References
Unless stated otherwise, all references are to the Programme and Abstracts of the 19th International AIDS Society World AIDS Conference, 22–25 July 2012, Washington DC. http://www.aids2012.org.
Human rights, access to treatment, activism and politics
Please see URL links included in this sections for details of each speaker and session.
New drugs and formulations
Gallant J et al. Cobicistat versus ritonavir as pharmacoenhancers in combination with atazanavir plus tenofovir disoproxil fumarate/emtricitabine: week 48 results. Oral abstract TUAB0103.
http://pag.aids2012.org/Abstracts.aspx?SID=202&AID=13085
Elion R et al. Efficacy and safety results from a randomized, double blind, active controlled trial of elvitegravir (once-daily) versus raltegravir (twice-daily) in treatment-experienced HIV-positive patients: long term 96-week data. Oral abstract TUAB0105.
http://pag.aids2012.org/Abstracts.aspx?SID=202&AID=5823
Raffi F et al. Once-daily dolutegravir (DTG; S/GSK1349572) is non-inferior to raltegravir (RAL) in antiretroviral-naive adults: 48 week results from SPRING-2 (ING113086). Late breaker oral presentation THLBB04.
http://pag.aids2012.org/abstracts.aspx?aid=20990
Nachman S et al. IMPAACT P1066: raltegravir (RAL) safety and efficacy in HIV+ youth two to 18 years of age through week 48. Oral Abstract TUAB0205.
http://pag.aids2012.org/flash.aspx?pid=1371
Hazra R et al. Pharmacokinetics, safety and efficacy of dolutegravir (DTG; S/GSK1349572) in HIV-1-positive adolescents: preliminary analysis from IMPAACT P1093. Oral abstract TUAB0203.
http://pag.aids2012.org/flash.aspx?pid=1307
Wang F et al. The HIV NRTI BMS-986001 does not degrade mitochondrial DNA in long term primary cultures of cells isolated from human kidney, muscle and subcutaneous fat. Poster abstract TUPE042.
http://pag.aids2012.org/abstracts.aspx?aid=17957
Guha M et al. Absence of renal and bone toxicity in non-clinical studies of BMS-986001, a nucleoside reverse transcriptase inhibitor (NRTI) of human immunodeficiency virus (HIV). Poster abstract TUPE041.
http://pag.aids2012.org/abstracts.aspx?aid=16832
Other studies
Mills A et al. Once-daily maraviroc in combination with ritonavir-boosted atazanavir in treatment-naïve patients infected with CCR5-tropic HIV-1 (study A4001078): 96-week results. Oral abstract TUAB0102.
http://pag.aids2012.org/Abstracts.aspx?SID=202&AID=16304
Poinsignon Y et al. Complicated atazanavir-associated cholelithiasis: a report of eight documented cases among 11 cases. Poster abstract MOPE099.
http://pag.aids2012.org/Abstracts.aspx?AID=6375
Rockstroh J et al. Long-term safety and efficacy of raltegravir (RAL)-based versus efavirenz (EFV)-based combination therapy in treatment-naïve HIV-1-infected patients: final 5-year results. Poster abstract LBPE19.
http://pag.aids2012.org/abstracts.aspx?aid=21410
Eron J et al. Final five-year results of the BENCHMRK studies: sustained antiretroviral effect of raltegravir and exploratory analysis of late outcomes based on early virologic response. Poster abstract TUPE025.
http://pag.aids2012.org/abstracts.aspx?aid=12576
Lee FJ et al. Skeletal muscle toxicity associated with raltegravir-based combination antiretroviral therapy in HIV-infected adults. 14th IWCADR, 19-21 July 2012, Washington. Oral abstract 015. Antiviral therapy 2012; 17 Suppl 2:A13. See:
Dinh M et al. HIV-1 female-to-male sexual transmission: evaluation of circumcised and uncircumcised penile tissue. Oral abstract MOLBA03.
http://pag.aids2012.org/abstracts.aspx?aid=21033
Reports launched at IAS
Please see URL links to each report included in this section.
Film reviews:
How to Survive a Plague, director David France.
http://www.howtosurviveaplague.com
“United in Anger: A history of ACT-UP”, director Jim Hubbard.
http://www.actuporalhistory.org/interviews/index.html
Glossary
abstract – a short summary of a study (usually about 350 words).
antiretrovirals – common name for all HIV drugs.
ARVs – abbreviation for ‘antiretrovirals’.
boosted-PI – a protease inhibitor taken with a booster dose of ritonavir.
CCR5 inhibitor – a type of HIV drug that stops HIV entering a CD4 cell.
CD4 cells – a type of lymphocyte (white blood cell). They are an important part of the immune system. CD4 cells are sometimes called T cells.
functional cure – when someone is still HIV positive but can stop ARVs without the virus causing any more harm.
integrase inhibitor – a type of HIV drug that works by stopping HIV from being integrated into the CD4 cell’s DNA (genetic material).
late breaker – last minute study results – like ‘late-breaking news’.
latent – not active at present, resting. Latently infected CD4 cells are CD4 cells that are infected with HIV but which are resting. HIV drugs do not work on resting cells.
latent reservoir – HIV-infected CD4 cells that are resting and not reached by ARV treatment.
prophylaxis – taking a drug to protect you against a potential illness.
protease inhibitor – a type of HIV drug that work but stopping new virus being able to leave a CD4 cell.
randomised – when neither you or your doctor chose which group you join in a research study.
sterilising cure – where every trace of HIV is removed from the body.

