The tuberculosis vaccine pipeline
30 June 2013. Related: Pipeline report, TB coinfection, Vaccines and microbicides.
Where are we going, where have we been?
By Mike Frick
Since the 2012 Pipeline Report, results from phase II trials and advances in preclinical development have brought the pipeline for new TB vaccines into sharper focus, even as correlates of protective immunity against TB remain elusive.
Nearly empty in 2000, the current pipeline includes 14 vaccine candidates in clinical trials and over 35 candidates in discovery or preclinical development. Compared to where the TB community stood just 10 years ago, the present pipeline attests to the reinvigorated investment in TB vaccine research and development over the last decade.
Yet disappointing results from the phase IIb efficacy trial of vaccine candidate MVA85A in infants as a boost to neonatal BCG announced in February 2013 require that we reevaluate the strategies that have brought us this far, paying critical attention to gaps in our knowledge of how Mycobacterium tuberculosis (MTB) interacts with the immune system and to the metrics we use to advance vaccine candidates through the pipeline.
Where have we been? The last 45 years
Forty-five years elapsed between the phase IIb trial of vaccine candidate MVA85A and the last TB vaccine efficacy trial, which evaluated the effectiveness of the bacille Calmette-Guérin (BCG) vaccine in Chengalpattu, India, in 1968. Over the decades, BCG became the most widely administered vaccine in the world, given over one billion times at low cost. Created by weakening strains of Mycobacterium (M.) bovis (the bacterium that causes TB in cattle), BCG was first introduced in 1921 and remains the only licensed vaccine for tuberculosis. [1] While BCG protects children against meningeal and disseminated forms of TB disease and death, it offers adults and adolescents highly variable protection against pulmonary TB, the form of the disease responsible for the vast majority of transmission and TB-related morbidity and mortality. As a live attenuated vaccine, BCG also poses a risk of leading to disseminated TB disease in people living with HIV and other immunosuppressive conditions. [2]
There remains a pressing need to develop superior vaccines against TB given the incomplete protection BCG offers and the safety risks it poses to immune-compromised individuals. A safe, effective vaccine against TB would provide a powerful tool for achieving zero TB deaths, new infections, and suffering, a goal endorsed by over 500 individuals and organizations since November 2012. [3] A vaccine that achieves complete elimination of MTB after infection, or offers complete protection against developing active TB, would offer the most direct path to zero. [4] Whether any of the vaccines under development can provide complete protection against TB remains unknown; yet the diversity of candidates in the pipeline could illuminate the degrees of protection afforded by different approaches.
Current TB vaccine candidates reflect several immunization strategies:
- Prime: Replace BCG with either live recombinant BCG (rBCG) or genetically attenuated MTB vaccines that confer greater safety and protective efficacy.
- Prime-boost: Boost the limited immunity conferred by BCG (or boost specific antigens presented by recombinant BCG or attenuated MTB) using either viral-vectored or adjuvanted subunit vaccines. Vaccines of this type would be administered as boosters to a BCG prime in infancy or in adolescence when BCG’s protection begins to dissipate.
- Immunotherapeutic: Develop therapeutic vaccines that might synergize with chemotherapy to shorten treatment for active TB disease or latent tuberculosis infection (LTBI). The therapeutic vaccines under development include whole-cell and fragmented mycobacteria, although several of these candidates also demonstrate prophylactic potential.
Common to these strategies is a focus on achieving cell-mediated immunity by inducing Th1 cytokines (e.g., IFNγ, TNFα, and IL-2) produced by either CD4 or CD8 T cells. These cytokines activate other cells capable of inhibiting the growth of MTB. [5, 6] This approach differs from the majority of licensed vaccines, which work primarily by inducing humoral immunity: antibodies produced by B cells. Observations of how MTB interacts with both human and animal immune systems have led researchers to assess the immunogenicity of vaccine candidates by measuring Th1 cytokines associated with CD4 T-cell activity. Deficiencies in IFNγ and other Th1 cytokines appear to place MTB-infected individuals at increased risk of developing TB disease. The strong correlation between CD4 T-cell depletion and higher risk of TB among people with HIV has also focused attention on the cellular immune response. [7]
A truism among TB vaccine developers is that without clear correlates of immunity, testing the efficacy of potential vaccines requires advancing the most promising candidates to large, expensive phase III clinical trials. The aperture of the pipeline, however, opens only so wide, so testing one candidate means delaying others due to limited financial resources and the small number of research sites equipped to host large clinical trials. Biomarkers that signal protection against primary infection and reactivation of TB disease could serve as surrogate endpoints, enabling shorter trials and more rational selection of candidates in the preclinical and early stages of clinical development. Yet biomarkers of protective immunity remain elusive, as do other important features of the dynamic human immune response to MTB infection.
The complexity of TB disease suggests that there is no single, silver-bullet biomarker of efficacy. Figure 1 illustrates where different biomarkers might fall in the immunologic life cycle of TB. Discovery of each would carry different implications for TB vaccine development. The ideal vaccine would offer protection across all populations and stages of TB’s immunologic life cycle. It would prevent infection of healthy individuals, provoke the immune system to clear infection in a person recently exposed to MTB, and prevent reinfection and progress toward active disease. [8] If different biomarkers correlate with different stages of infection and disease, we may need multiple vaccines that protect distinct target populations through separate immunologic mechanisms. The absence of biomarkers of protective immunity has exerted a profound impact on the shape, composition, and strategies that define the current pipeline.
Figure 1: What do we talk about when we talk about biomarkers? Biomarkers for different stages of MTB’s immunologic life cycle
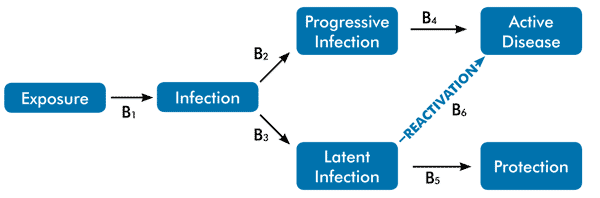
B = biomarker. Prospective cohort studies may help to differentiate biomarkers of active disease (B4) from latent infection (B5). An open question remains whether such cohorts can distinguish markers of reactivated disease (B6) from markers signaling the progression from initial infection to active disease (B2, B4). Human mycobacterial challenge models may help to identify biomarkers characterizing the transition from exposure to infection (B1). Several preclinical studies in animals are exploring what enables 90 percent of those latently infected to resist disease (B5). For example, studies in nonhuman primates are investigating the diverse activity characterizing granuloma formation and sterilization within a single host. [9] While these approaches may identify candidate biomarkers, only successful phase III trials can serve to validate biomarkers as surrogate endpoints for subsequent studies.
Image adapted from Ottenhoff TH, Ellner JJ, Kaufmann SH. Ten challenges for TB biomarkers. Tuberculosis (Edinb). 2012 Mar;92 Suppl 1:S17–20.
Where are we now? The clinical TB vaccine pipeline
There are currently 14 vaccine candidates undergoing or preparing to enter human clinical trials. Four of these candidates have reached phase I trials to demonstrate safety, and eight have reached phase II trials to assess their safety and immunogenicity. M. vaccae and M. indicus pranii, two whole-cell mycobacteria vaccine candidates, have completed phase III trials.
| Agent | Strategy | Type | Sponsors | Status |
|---|---|---|---|---|
| M. indicus pranii | Immunotherapeutic | Whole-cell M. indicus pranii | Department of Biotechnology (Government of India), Cadila Pharmaceuticals | Phase III |
| M. vaccae | Immunotherapeutic | Whole-cell M. vaccae | AnHui Longcom | Phase III pending |
| MVA85A/AERAS-485 | Prime-boost | Viral vector | Oxford University, Aeras | Phase IIb |
| M72 + AS01 | Prime-boost | Adjuvanted subunit | GSK, Aeras | Phase IIb |
| Crucell Ad35/ AERAS-402 | Prime-boost | Viral vector | Crucell, Aeras | Phase II (formerly phase IIb) |
| VPM1002 | Prime | Live recombinant rBCG | Vakzine Projekt Management GmbH, Max Planck Institute for Infection Biology, TuBerculosis Vaccine Initiative (TBVI), Serum Institute of India | Phase IIa |
| RUTI | Immunotherapeutic | Fragmented MTB | Archivel Farma | Phase IIa |
| Hybrid 1 + IC31 | Prime-boost | Adjuvanted subunit | Statens Serum Institut (SSI), TBVI, Intercell, European & Developing Countries Clinical Trials Partnership | Phase IIa |
| Hybrid 56 + IC31 | Prime-boost | Adjuvanted subunit | SSI, Aeras | Phase IIa |
| Hybrid 4 + IC31/AERAS-404 | Prime-boost | Adjuvanted subunit | Aeras, Sanofi Pasteur | Phase IIa |
| ID93 + GLA-SE | Prime-boost | Adjuvanted subunit | Infectious Disease Research Institute, Aeras | Phase I |
| Ad5Ag85A | Prime-boost | Viral vector | McMaster University, CanSino | Phase I |
| MTBVAC | Prime | Live genetically attenuated MTB | University of Zaragoza, Biofabri, TBVI | Phase I |
| Dar-901 | Prime-boost | Whole-cell M. vaccae | Geisel School of Medicine at Dartmouth University | Phase I pending |
Note: For each candidate under the prime-boost strategy, trials are evaluating the experimental vaccine in the left-hand column given as a boost to a BCG prime.
Live Recombinant Vaccine Candidates
Two live vaccine candidates designed to replace BCG remain in the pipeline: VPM1002 and MTBVAC. As a live recombinant form of BCG, VPM1002 aims to overexpress key MTB antigens. By contrast, MTBVAC contains live MTB weakened to become less virulent while still provoking a cellular immune response. A phase I trial of an earlier live recombinant candidate, AERAS-422, ended in 2011 due to adverse events (reactivation of shingles). Another live recombinant candidate, rBCG30, completed a phase I trial in 2004 but has been placed on hold without further development plans. [10]
VPM1002
Currently completing a phase IIa trial, VPM1002 remains the most advanced live recombinant BCG vaccine candidate. Lead developers for VPM1002 include Vakzine Projekt Management GmbH, the Max Planck Institute for Infection Biology, and the TuBerculosis Vaccine Initiative (TBVI). Vakzine Projekt Management GmbH recently formed a partnership with the Serum Institute of India to support VPM1002’s development. [11] The phase IIa trial hosted by Stellenbosch University in South Africa enrolled its final participant in May 2012. The trial will compare the safety of VPM1002 and BCG in 48 BCG-naive, HIV-negative newborns. Preliminary data suggest the vaccine is safe and stimulates an immune response similar to an equivalent dose of BCG, as measured by CD4 and CD8 T-cell activity. [12]
A second phase IIa trial will begin by early 2014 and will assess the safety and immunogenicity of VPM1002, compared with BCG, in HIV-exposed and -unexposed newborns. [13] VPM1002 remains one of the few candidates with an infant or newborn product-development profile at a time when the pipeline is shifting toward vaccine development for adolescents and adults based on data showing that older age groups account for the majority of pulmonary TB transmission.
MTBVAC
A phase I trial of MTBVAC, a live attenuated MTB vaccine candidate, began in Lausanne, Switzerland, in January 2013. MTBVAC is the first live vaccine constructed from attenuated MTB to enter clinical trials and is being developed by the University of Zaragoza in partnership with Biofabri, a Spanish pharmaceutical company, and TBVI. [14] The trial will enroll 36 BCG-naive, HIV-negative adult volunteers to compare the tolerability and safety of MTBVAC with BCG. Since MTBVAC is a live vaccine, the initial series of trials will take place in HIV low-incidence settings.
MTBVAC contains MTB attenuated by the deletion of two virulence genes: phoP and fadD26. Analyses of clinical isolates from an M. bovis XDR strain (B strain) responsible for an MDR-TB outbreak in Spain in the early 1990s suggest that phoP plays an important role in regulating mycobacterial lipids that contribute to MTB virulence. [15, 16] Preclinical studies indicate that MTBVAC is safe in immune-compromised mice and protects guinea pigs and nonhuman primates against MTB infection. Notably, vaccination with MTBVAC in mice resulted in greater differentiation of CD4 T-cells into effector and memory T-cells compared with BCG. [17] This finding suggests that MTBVAC has the potential to confer longer-lasting immunity than BCG.
Viral Vector Vaccine Candidates
Viral vector vaccines use weakened, nonreplicating viruses to transport MTB DNA into human cells, where the DNA is transcribed into proteins that provoke an immune response. The three viral vector candidates in human trials represent the most advanced branch of the pipeline, with two of these undergoing multiple phase II trials and the third recently completing an initial phase I study. Each intends to boost the effects of earlier vaccination with BCG under a prime-boost strategy.
MVA85A/AERAS-485
The most significant news from the TB vaccines world in the past year came from the disappointing results of the phase IIb trial of vaccine candidate MVA85A/AERAS-485 in BCG-primed infants. This marked the first efficacy trial of an engineered TB vaccine since the Chengalpattu, India, trial of BCG in 1968. Developed by the Oxford-Emergent Tuberculosis Consortium with support from Aeras, MVA85A is a recombinant strain of modified vaccinia virus Ankara (MVA) that expresses MTB antigen Ag85A. MVA85A aims to boost earlier vaccination with BCG, as most BCG-vaccinated individuals carry immunologic memory of the Ag85A antigen.
The phase IIb trial randomized 2,794 BCG-vaccinated, HIV-negative infants ages four to six months to receive either MVA85A or placebo (Candida skin test antigen). The primary study endpoint was safety with two secondary efficacy outcomes: protection against TB disease and prevention of MTB infection. Although admirably safe, MVA85A did not protect infants against either TB disease or MTB infection. The study team reported 39 cases of incident TB in the placebo group compared with 32 cases in the vaccine group, for an overall estimated vaccine efficacy of 17.3 percent. For the MTB infection endpoint, 171 infants in the placebo group and 178 infants in the MVA85A group became infected with MTB during the study, yielding an estimated efficacy against infection of –3.8 percent. The differences between vaccine and placebo groups with respect to either secondary efficacy endpoint were not statistically significant. [18]
Although disappointing, results from the MVA85A trial may inform the development of other candidates in the pipeline. MVA85A stimulated CD4 T cells to produce the cytokines IFNγ, TNFα, and IL-2 in vaccinated infants, but only at modest levels that did not add protection to BCG. This response was lower than that predicted by animal studies, highlighting the incomplete window into efficacy offered by current animal models. Analyses of a sample bank collected during the trial may also help to identify correlates of risk of TB disease. [19]
Helen McShane, professor of vaccinology at Oxford University and lead developer of MVA85A, raised the possibility that MVA85A may afford greater protection to adolescents and adults. Adults vaccinated with BCG have demonstrated stronger responses to Ag85A compared with infants. [20] A second phase IIb trial of MVA85A among BCG-vaccinated adults living with HIV in South Africa and Senegal should help to answer this question; it is continuing to enroll participants. [21]
Crucell Ad35/AERAS-402
More disappointing news surrounds Crucell Ad35/AERAS-402, another viral vector vaccine candidate that entered phase IIb trials, one enrolling BCG-vaccinated infants and the other enrolling BCG-vaccinated, HIV-positive adults. Crucell Ad35 uses human adenovirus 35 expressing three MTB antigens: Ag85A, Ag85B, and TB10.4. Immunogenicity data from earlier phase I trials indicated that Crucell Ad35 elicited a robust CD8 T-cell response, but only a modest response from CD4 T cells and their associated cytokines. [22] After reviewing preliminary data, each trial was revised from a phase IIb proof-of-concept study to a smaller phase II study, with safety and immunogenicity as the primary endpoints and without enrollment of the larger study population needed to evaluate efficacy. Without the efficacy groups, the trial will now enroll 500 participants instead of the 4,000 specified by the original protocol. [23] This recategorization has dimmed the prospects of Crucell Ad35, and reflects low immunogenicity response rates to the vaccine in the early, blinded data.
Ad5Ag85A
Like MVA85A, vaccine candidate Ad5Ag85A aims to boost BCG by overexpressing the MTB antigen 85A. Ad5Ag85A is a recombinant, replication-deficient adenovirus serotype 5 vaccine vector and was recently evaluated in a phase I trial that enrolled 24 HIV-negative adults: 12 previously vaccinated with BCG and 12 who were BCG-naive. [24] The study reported a few mild adverse reactions at the injection site, all resolved within 24 hours, and no vaccine-related serious adverse events. The vaccine showed greater immunogenicity in the study group primed with BCG, activating cytokine production in both CD4 and CD8 T cells. Volunteers in the BCG-naive group also responded to the vaccine, although at a slower rate.
McMaster University, where the vaccine was conceptualized, plans to conduct a phase I trial evaluating aerosol delivery of Ad5Ag85A. [25] Some concerns surround the appropriateness of using the Ad5 vector to deliver vaccine in HIV-positive adults given a safety signal that emerged from the STEP trial evaluating a potential HIV vaccine built on the Ad5 platform. In the STEP trial, uncircumcised vaccine recipients with preexisting Ad5 antibodies showed an increased risk of HIV acquisition, although the mechanism of this reaction remains unknown. [26]
McMaster University is now developing Ad5Ag85A with support from CanSino, a Chinese biotechnology company based in Tianjin.
Adjuvanted Subunit Vaccine Candidates
The prime-boost strategy also includes several adjuvanted subunit vaccine candidates that contain fusions of different MTB protein antigens in combination with an adjuvant. Vaccines based on purified antigens are less immunogenic than live attenuated or inactivated whole-cell vaccines and thus benefit from adjuvants (pharmacological agents that boost the body’s immune response to antigens). Adjuvanted subunit vaccines represent the most well-populated segment of the pipeline, with five candidates in clinical development.
M72 + AS01
The most advanced adjuvanted subunit vaccine, M72 + AS01, is completing several phase IIa studies in preparation for a multisite phase IIb trial in Africa. M72 + AS01 contains a fusion protein of MTB antigens 32A and 39A in the adjuvant AS01. It remains one of just a few vaccines with backing from a major pharmaceutical company, GlaxoSmithKline (GSK), with additional support provided from Aeras.
Phase I/IIa trials suggest that M72 + AS01 has an acceptable safety profile and stimulates both CD8 and CD4 T-cell responses. The CD4 T-cell response has emerged as particularly interesting. In a phase IIa trial among 45 MTB-infected and -uninfected adults in South Africa, M72 + AS01 triggered T cells outside of the typical Th1 and Th17 responses seen in other vaccine candidates. These novel T-cell populations appeared to include Treg cells, which may mediate the inflammation caused by Th1 and Th17 cytokines. T-cell counts after administration of M72 + AS01 were also higher in MTB-infected participants than in individuals uninfected with MTB, suggesting that vaccination with M72 + AS01 may boost T-cell populations primed by natural MTB infection. This finding stands in contrast to a small adult trial of MVA85A, which showed no significant difference in magnitude of Ag85A-specific T cells across MTB-infected and -uninfected study participants after vaccination. [27]
The three ongoing or recently completed phase IIa studies aim to evaluate the safety and immunogenicity of M72 + AS01 in diverse patient populations: infants in Gambia; HIV-positive adults in Chennai, India; and adults with TB disease in Taiwan and Estonia. [28] The phase IIb study will be the largest adult trial of a novel TB vaccine, aiming to enroll 4,500 HIV-negative adult volunteers in TB endemic communities in sub-Saharan Africa. The primary endpoint will examine protective efficacy of two doses of M72 + AS01 against pulmonary TB disease. Secondary endpoints will include safety and immunogenicity, as well as an exploratory endpoint assessing the vaccine’s effectiveness at preventing MTB infection. [29]
Hybrid 1, Hybrid 56, and Hybrid 4 + IC31
The Statens Serum Institut (SSI) in Denmark is choosing between several adjuvanted subunit vaccines to advance into phase II efficacy trials.
Hybrid 1 + IC31 is beginning a phase IIa trial with backing from SSI, TBVI, and the European & Developing Countries Clinical Trials Partnership (EDCTP). Hybrid 1 + IC31 contains antigens Ag85B and ESAT6 in IC31, an adjuvant developed by Intercell. Phase I data indicate that Hybrid 1 + IC31 elicits a robust CD4 T-cell response as measured by IFNγ production. SSI also supported the Hybrid 1 candidate paired with the adjuvant CAF01 in a separate phase I trial. CAF01 will remain a backup adjuvant for Hybrid 1, with no plans for further evaluation. Selection between the IC31 and CAF01 adjuvants mainly reflects timing; CAF01 was not ready for testing in humans until much later than IC31. [30]
Although Hybrid 1 + IC31 remains under investigation in two studies, it will likely be phased out in favor of Hybrid 56 + IC31. Else Agger of SSI said that “Hybrid 56 + IC31 will stand on the shoulders of Hybrid 1 + IC31.” The Hybrid 56 + IC31 vaccine contains antigens expressed during both active TB disease (85B and ESAT6) and latency (Rv2660). The first phase I trial of this candidate began in South Africa in 2011 and completed enrollment in November 2012. Hybrid 56 + IC31 appears safe and well-tolerated, and a second phase I, or possible phase IIa, safety and dose-finding study will begin in May 2013 pending favorable immunogenicity results from the first trial. The phase I/IIa trial will take place in South Africa and one additional African site among MTB-uninfected and latently infected adults. [31] Hybrid 56 + IC31 appears more stable than Hybrid 1 + IC31 in pharmaceutical evaluations. Animal models also suggest that Hybrid 56 demonstrates a higher ESAT6 response than Hybrid 1 and has better vaccine efficacy during late-stage MTB infection, although the mechanisms of these reactions remain unclear. [32]
A separate fusion protein candidate, Hybrid 4 + IC31/AERAS-404, also uses adjuvant IC31 and is being developed by Aeras and Sanofi Pasteur following initial support from SSI. Hybrid 4 + IC31 fuses the antigens Ag85B and TB10.4. The TB10.4 antigen is expressed in both MTB and BCG, unlike the ESAT6 antigen used in Hybrid 1 + IC31 and Hybrid 56 + IC31, which is not present in BCG. ESAT6’s role as a common diagnostic reagent in several commercial tests for TB has encouraged researchers to reserve it for diagnostic use by replacing it with an antigen in the same gene family: TB10.4. [33, 34] Hybrid 4 + IC31 has completed four phase I studies in adults, including a study in 70 volunteers to assess how it performs as a booster to BCG in preparation for future phase II studies in infants. [35] A phase I/IIa safety and immunogenicity study in infants is expected to start in June 2013 at multiple sites in South Africa. [36]
ID93 + GLA-SE
ID93 + GLA-SE is an adjuvanted subunit vaccine in phase I trials that includes a latency antigen. ID93 + GLA-SE combines four MTB antigens, three expressed in active TB disease (Rv2608, Rv3619, Rv3620) and one expressed during latency (Rv1813). Testing in animals suggests that GLA-SE enhances both Th1 and Th2 immune responses. [37] Lead developers for ID93 + GLA-SE include Aeras and the Infectious Disease Research Institute in Seattle.
A phase I study evaluating ID93 + GLA-SE’s safety among BCG-naive adults is currently under way in the United States with a second study among BCG-primed adults planned in South Africa. The trial in the U.S. will enroll four cohorts in escalating doses of both adjuvant and antigen; each cohort will receive three immunizations spaced one month apart. The fourth cohort began enrollment in March 2013, and the investigators have reported no adverse events to date. [38]
In addition to these phase I trials, preclinical studies in mice and nonhuman primates are assessing whether ID93 + GLA-SE can serve as a therapeutic vaccine administered in conjunction with standard antimicrobial treatment. When given with isoniazid and rifampicin, ID93 + GLA-SE elicited a stronger Th1 immune response, shortened the length of chemotherapy, and extended survival time in mice and monkeys. Future preclinical work will evaluate ID93 + GLA-SE as a therapeutic adjunct against MDR-TB strains and paired with first-line drug regimens. [39]
Whole-Cell or Fragmented Mycobacteria Vaccine Candidates
Whole-cell mycobacteria vaccines represent an older branch of the pipeline, as two of these candidates had entered clinical trials by the early 1990s. Vaccines of this type contain inactivated, replication-deficient whole-cell or fragmented mycobacteria. Each of the whole-cell vaccine candidates discussed below may serve as therapeutic vaccines that would synergize with chemotherapy in order to improve treatment for either active TB disease or LTBI. In addition to their immunotherapeutic potential, each is also being studied under a more traditional prime-boost vaccination strategy.
RUTI
Vaccine candidate RUTI consists of fragmented MTB and is being developed by Archivel Farma, a Spanish biotechnology company, as a therapeutic vaccine to shorten treatment of both LTBI and active TB disease. In May 2011, RUTI completed a phase II trial in HIV-positive and HIV-negative people with LTBI. Results of the trial indicate that RUTI elicits an immune response by activating MTB antigens ESAT6 and Ag85B. Archivel Farma is currently looking for a financial partner for a planned phase III trial that will test a single dose of RUTI under two scenarios: first, as an adjunct to chemotherapy to prevent active TB disease in people with LTBI; and second, to prevent relapse episodes in active TB patients by administering the vaccine in the continuation phase of treatment. [40]
Preclinical studies have also assessed RUTI’s prophylactic effect in mice and guinea pigs. These findings raise two possibilities for RUTI’s development as a preventive vaccine: first, that RUTI might be administered to recently infected people who have negative TB skin tests but are contacts of index cases; and second, that RUTI might boost BCG under a more conventional prime-boost strategy. [41,42]
Dar-901
After a hiatus, the whole-cell mycobacteria vaccine SRL172 studied in the earlier phase III DarDar trial returns to the pipeline as Dar-901. Developed by the Geisel School of Medicine at Dartmouth University, Dar-901 consists of inactivated, whole-cell Mycobacterium vaccae. A new manufacturing method developed by Aeras represents the primary difference between Dar-901 and SRL172. The M. vaccae used in Dar-901 is broth-grown, a more scalable production method than the agar-grown M. vaccae used in the DarDar trial.
Dar-901 has completed preclinical testing in animals and will begin a phase I trial in the United States in late 2013 assessing the safety of three doses of vaccine in BCG-vaccinated, HIV-positive adults. [43]
Earlier work on this candidate culminated in the phase III DarDar trial that evaluated the protective effect of SRL172 against disseminated TB disease in HIV-positive, BCG-primed adults in Dar es Salaam, Tanzania. This trial stopped early due to slow accrual of disseminated TB cases following improved HIV interventions in the study population. [44]
In contrast to the DarDar study, the Dar-901 study will use definite, culture-confirmed TB disease as the primary endpoint rather than disseminated TB. If successful in phase I, further phase IIa studies will evaluate Dar-901 among both HIV-positive and -negative adults in Tanzania. [45]
Mycobacterium vaccae
AnHui Longcom, a Chinese pharmaceutical company, is studying M. vaccae as an adjunct to standard antimicrobial therapy. The company already holds a license from the Chinese State Food and Drug Administration to distribute M. vaccae as a therapy-shortening adjunct for pulmonary TB. A phase IIb trial among people with MTB infection in Nanjing recently ended, and the company plans to begin a phase III trial soon. [46] A 2003 Cochrane Collaboration review found no evidence that M. vaccae immunotherapy benefits patients with pulmonary TB as measured by either mortality or treatment duration, a conclusion that calls into question AnHui Longcom’s decision to conduct additional efficacy trials. [47] None of the clinical trials conducted by AnHui Longcom since the Cochrane review has been published in English-language, peer-reviewed journals.
Mycobacterium indicus pranii
A more unconventional path charts the development of Mycobacterium indicus pranii (MIP; also called Mycobacterium w), a fast-growing, nonpathogenic mycobacterium first developed as a vaccine against leprosy. MIP shares B- and T-cell epitopes (the specific part of an antigen that the cells recognize) with both Mycobacterium leprae and MTB, an observation that led the Indian Department of Biotechnology (DBT) to study its potential as a TB vaccine by revisiting data from the original leprosy trials.
Instead of launching a new clinical trial, the government of India nested a retrospective, observational analysis of MIP’s protective effect against TB disease among the 28,948 people enrolled in the phase III trial of MIP against leprosy. After resurveying this population 10 years after the initial trial, investigators found fewer cases of TB disease among those vaccinated with MIP compared with the unvaccinated control group. [48] However, the retrospective nature of this study left investigators unable to calculate measures of vaccine efficacy or establish a causal link between MIP vaccination and lower risk of developing pulmonary TB.
Consequently, investigators at the DBT are now working in reverse order by returning to animal studies to assess MIP’s immunogenicity against MTB. Preclinical investigations are assessing both live and killed formulations of MIP, as well as a novel aerosol route of delivery. [49] Immunogenicity data from these studies indicate that MIP stimulates strong Th1 and Th17 immune responses through both CD8 and CD4 T-cell cytokines. [50]
The Drug Controller of India has already licensed MIP for use in humans; the product has been evaluated in three recently completed phase III trials sponsored by the DBT and Cadila Pharmaceuticals. These trials evaluated a killed formulation of MIP as an adjunct to first-line antimicrobial therapy among category I TB patients, category II TB patients, and individuals with tuberculous pericarditis, respectively. [i] Results have yet to be published.
Where are we going? Recommendations on getting to zero with vaccines
A vaccine that successfully acts against pulmonary TB would offer a powerful, and possibly essential, tool for reaching zero tuberculosis deaths, new infections, and suffering. Mathematical modeling commissioned by Aeras suggests that a 60 percent–effective vaccine given to adolescents could avert 71 percent (67 million) of the TB cases and 60 percent (8 million) of the TB deaths projected to occur between 2014 and 2050. [51] However, researchers must clear several hurdles on the road to an efficacious TB vaccine, beginning with unanswered questions of TB immunology. The following recommendations outline priority areas for research and discovery.
1. Prioritize the science behind biomarker discovery.
Biomarkers of protective immunity against TB are urgently needed to reduce the cost, time, and uncertainty of advancing candidates through the pipeline. Discovery of potential biomarkers will not refashion the clinical pipeline overnight. Even with biomarkers, phase III trials will still be required to determine vaccine efficacy, and markers of protective immunity themselves will require clinical validation. Yet, validated biomarkers would allow researchers to look for efficacy earlier and improve the selection of candidates for expensive late-phase trials. Launching numerous efficacy trials without a clearer picture of protective immunity risks inducing research fatigue among host countries, donors, policy makers, and TB-affected communities. The cost these endeavors pose to health systems and communities cannot be overstated. Stefan Kaufmann has estimated that even in areas with high TB incidence, phase III trials may need to include 20,000 people per study arm to obtain statistically significant findings. [52]
Ultimately, biomarkers are tools whose discovery will follow scientific research unveiling the dynamics of how the human immune system responds to MTB at different stages of infection and disease. The field’s focus on these tools should not jump ahead of the advances in microbiology, immunology, and other disciplines that will make this search possible. Just as no single biomarker will produce an efficacious vaccine, no strategy pursued in isolation will uncover correlates of immunity. The research areas described below may offer complementary insights into biomarker discovery.
- (a) Correlates of risk. One method seeks to identify correlates of risk for TB disease through observational cohort studies. Willem Hanekom of the South African TB Vaccine Initiative is following two prospective cohorts to identify biomarkers distinguishing MTB-infected adolescents and infants who develop active TB disease from those who do not. Preliminary analyses of blood samples from the adolescent cohort have identified more than 1,200 genes differentially expressed between adolescents who control MTB infection and those who develop active disease. Genes regulating myeloid cell inflammation appear to play an important role. [53, 54]
- (b) Human challenge model. Development of a human mycobacterial challenge model may also illuminate correlates of risk and protective immunity. The most obvious route to a human challenge model for TB remains untenable, since exposing people to virulent MTB poses grave ethical concerns. Helen McShane is leading several studies to develop a human mycobacterial challenge model that uses intradermal BCG vaccination as a surrogate for aerosol MTB infection. [55, 56]
- (c) Host/pathogen interaction. The information conveyed by biomarkers will likely be contextual, defined by both the stage of MTB infection or TB disease under study and the scientific questions asked by vaccine researchers. Given the importance of context, several questions related to the interaction of host and pathogen deserve increased attention.
First, the immunologic life cycle of MTB is poorly understood, and although 90 percent of infected individuals resist active disease, the mechanisms of this resistance remain opaque.
Second, our window into the cellular immune response required to overcome active TB disease is too narrow. Induction of Th1 cytokines appears necessary but not sufficient for conferring protective immunity. The role of humoral immunity through B cells and antibodies also remains unclear.
Third, our assumptions based on markers observed in animal studies have not translated into human studies. No animal model has been validated with human disease, and few models have tested candidate vaccines against the range of MTB strains observed in the field.
3. Pursue innovation within clinical trials.
Clinical trials should synergize with advances in basic science through an iterative process of research and discovery. In Tuberculosis Vaccines: a Strategic Blueprint for the Next Decade, Aeras and TBVI outlined stage-gate criteria for advancing vaccines from one stage of the pipeline to another. These criteria evaluate candidates in nine categories, ranging from safety to regulatory strategy. [57] Yet the immunogenicity and efficacy criteria appear underdeveloped compared with those for the other categories, perhaps reflecting the distance untraveled in basic science. Refining the immunogenicity criteria would endow the stage gates with greater utility for advancing only the most promising candidates.
To enable this refinement, clinical trials should collect serum and cell samples that can be retrospectively analyzed to search for biosignatures distinguishing participants who demonstrate different outcomes or vaccine response. Sample-bank analyses should be back-translated to move preclinical models closer to mirroring human TB infection and disease. This potential could be maximized by linking samples collected by different trials to larger biobanks, or by making these data publicly available to other research teams. The diversity of TB disease across human populations means that analyses limited by geography may elide key insights into the variability of host/pathogen interaction. The establishment of integrated biobanks faces serious logistical challenges stemming from protocol differences across trials. The harmonization of trials would enable clinical research to better support basic science.
Researchers should map innovative pathways within the current strategies that define the clinical pipeline. Over a dozen vaccine candidates have entered clinical trials since the turn of the century, yet each has charted parallel, nonintersecting research trajectories. Combining different candidates might offer one way to capture the respective strengths of different vaccines under development through a twist on the dominant prime-boost strategy.At the Third Global Forum on TB Vaccines, Helen McShane raised the possibility of combining MVA85A and Crucell Ad35, as these candidates target distinct MTB antigens and elicit CD4 and CD8 T-cell responses, respectively. The prospect of combination trials raises an important regulatory question: will vaccines used in combination need to retrace all of the steps stretching from preclinical to clinical trials together if they have completed these stages individually? The role of BCG in such an approach also deserves attention. Instead of maintaining BCG as the prime inoculation, should vaccination with BCG be seen as the background on top of which researchers prime and boost with a combination of different vaccines?
3. Increase funding for TB vaccine research, including basic science.
In 2011, donors spent $95.4 million on TB vaccine R&D, a sum that represents only 25 percent of the annual investment of US$380 million called for in the Stop TB Partnership’s Global Plan to Stop TB 2011–2015. [58] Additional funding alone will not produce an effective vaccine; money must be strategically placed to solve intractable issues slowing TB vaccine R&D. The National Institute of Allergy and Infectious Diseases has committed US$10 million to fund Tuberculosis Research Units investigating the biology of MTB infection and TB disease. This offers one example of targeting funding toward research with the potential to identify biomarkers.
Conclusion
Despite well-populated clinical and preclinical pipelines, the TB vaccine community faces a curious catch-22: without known correlates of protective immunity, many believe that efficacy trials offer the only route for evaluating whether candidate vaccines prevent TB disease. But trials that demonstrate no statistically significant protective efficacy against TB cannot illuminate the correlates of immunity that would revolutionize the field. Markers of protective immunity are not an end in themselves, but instead offer a suite of tools, discovery of which will depend on major advances in microbiology and immunology. Extricating ourselves from this quandary will require pairing a renewed dedication to basic science with a willingness to challenge the assumptions underlying current models.
| Antigengene name (protein name) | Stage of expression | Present in which vaccine candidates |
|---|---|---|
| Rv3804 (Ag85A) | Active disease | MVA85A, Crucell Ad35, Ad5Ag85A |
| Rv1886 (Ag85B) | Active disease | Hybrid 4 + IC31, Hybrid 56 + IC31, Crucell Ad35 |
| Rv3875 (ESAT6) | Active disease | Hybrid 1 + IC31, Hybrid 56 + IC31 |
| Rv0288 (TB10.4) | Active disease | Hybrid 4 + IC31, Hybrid 1 + IC31, Crucell Ad35 |
| Rv1196 (Mtb39A) | Active disease | M72 + AS01 |
| Rv0125 (Mtb32A) | Active disease | M72 + AS01 |
| Rv2608 | Active disease | ID93 + GLA-SE |
| Rv3619 | Active disease | ID93 + GLA-SE |
| Rv3620 | Active disease | ID93 + GLA-SE |
| Rv1813 | Latent infection | ID93 + GLA-SE |
| Rv2660 | Latent infection | Hybrid 56 + IC31 |
Acknowledgments
I would like to thank Christine Sizemore, Richard Jefferys, Erna Balk, and Jennifer Woolley for providing helpful reviews of an early draft.
Endnotes
- McShane, H. Tuberculosis vaccines: beyond bacilli Calmette-Guérin. Philos Trans R Soc Lond B Biol Sci. 2011 Oct 12;366(1579):2782–9. doi: 10.1098/rstb.2011.0097.
- Colditz G, Brewer T, Berkey C, et al. Efficacy of BCG vaccine in the prevention of tuberculosis. Meta-analysis of the published literature. JAMA. 1994;271(9):698–702. doi: 10.1001/jama.271.9.698.
- Treatment Action Group. The zero declaration: zero TB deaths, new infections, and suffering. New York: Treatment Action Group; 2012. Available from:
http://www.treatmentactiongroup.org/tb/advocacy/zero. (Accessed April 10, 2013) - Ottenhoff, T, Kaufmann, S. Vaccines against tuberculosis: where are we and where do we need to go? PLoS Pathogens. 2012;8(5):e1002607. doi: 10.1371/journal/ppat.1002607.
- Evans T, Brennan M, Barker L, et al. Preventive vaccines for tuberculosis. Vaccine. 2013;31(S):B223–226. doi: 10.1016/j.vaccine.2012.11.081.
- Schluger N, Rom W. The host immune response to tuberculosis. Am J Respir Crit Care Med. 1998;157(3):679–91. doi: 10.1164/ajrccm.157.3.9708002.
- Walzl G, Ronacher K, Hanekom W, Scriba TJ, Zumla A. Immunological biomarkers of tuberculosis. Nat Rev Immunol. 2011 May;11(5):343–54. doi: 10.1038/nri2960.
- Cayabyab M, Macovei L, Campos-Neto A. Current and novel approaches to vaccine development against tuberculosis. Front Cell Infect Microbiol. 2012;2:154. doi: 10.3389/fcimb.2012.00154.
- Flynn J. Animal models of clinically latent infection. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Ottenhoff, T, Kaufmann, S. Vaccines against tuberculosis.
- Grode, Leander. (Vakzine Projekt Management GmbH, Hannover, Germany). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 26.
- Grode L. Latest development of VPM1002: a new prime vaccine on the horizon. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Ibid.
- Martin C. MTBVAC, from the lab to the clinical trials. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Martin C, Williams A, Hernandez-Pando R, et al. The live Mycobacterium tuberculosis phoP mutant strain is more attenuated than BCG and confers protective immunity against tuberculosis in mice and guinea pigs. Vaccine. 2006;24(17):3408–19. doi: 10.1016/j.vaccine.2006.03.017.
- Soto A, Mene M, Samper S, Go A, Barcia M, Martin C. IS6100 mediates increased transcription of the phoP virulence gene in multidrug-resistant clinical isolate responsible for tuberculosis outbreaks. J Clin Microbiol. 2004 Jan;42(1):212–9. doi: 10.1128/JCM.42.1.212-219.2004.
- Martin C. MTBVAC, from the lab to the clinical trials. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Tameris M, Hatherhill M, Landry B, et al. Safety and efficacy of MVA85A, a new tuberculosis vaccine, in infants previously vaccinated with BCG: a randomized, placebo-controlled phase 2b trial. Lancet. 2013;381(9871):1021–8. doi: 10.1016/S0140-6736(13)60177-4.
- McShane H. Boosting BCG with MVA85A – clinical trials and efficacy data. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Ibid.
- McShane, Helen. (Oxford University, Oxford, England). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 27.
- Douoguih M. AERAS-402/Crucell Ad35. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Barker, Lew (Aeras, Rockville, MD). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 30.
- Smaill F. Potent T cell immunogenicity of a novel human type 5 adenovirus-based tuberculosis vaccine in humans despite pre-existing anti-adenovirus 5 immunity. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Smaill, Fiona (McMaster University, Hamilton, Ontario). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 26.
- Jefferys R. The role of Ad5-specific CD4 T-cells in enhancing risk of HIV acquisition in the Merck vaccine trial. Michael Palm HIV basic science, vaccines, and prevention project weblog. 2009 July 22. Available at:
http://tagbasicscienceproject.typepad.com (Accessed 2013 May 2). - Day C, Tameris M, Mansoor N, et al. Induction and regulation of T cell immunity by the novel TB vaccine M72/AS01 in South African adults. Am J Respir Crit Care Med. 2013 Jan 10. doi:10.1164/rccm.201208-1385OC. [Epub ahead of print]
- Tait D. Progress of the M72/AS01E tuberculosis vaccine candidate into phase IIb efficacy trial. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Tait, M72/AS01E tuberculosis vaccine candidate.
- Agger E. Prospects for novel tuberculosis protein-based subunit vaccines. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Ibid.
- Agger, Else (Statens Serum Institut, Copenhagen, Denmark). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 29.
- Davila J, McNamara L, Yang Z. Comparison of the predicted population coverage of tuberculosis vaccine candidates Ag85B-ESAT-6, Ag85B-TB10.4, and Mtb72f via a bioinformatics approach. PLoS One. 2012;7(7):e40882. doi: 10.1371/journal.pone.0040882.
- Billeskov B, Elvang T, Anderson P, et al. The HyVac4 subunit vaccine efficiently boosts BCG-primed anti-mycobacterial protective immunity. PLoS One. 2012;7(6): e39909. doi: 10.1371/journal.pone.0039909.
- Pantaleo G. A phase I double-blind, randomized, placebo-controlled trial to evaluate the safety and immunogenicity of BCG and AERAS-404 administered as a prime-boost regimen to HIV-negative, TB-negative, BCG-naïve adults. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Woolley, Jennifer. (Aeras, Rockville, MD). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 24.
- Reed S. ID93. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Coler, Rhea (Infectious Disease Research Institute, Seattle, WA). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 27.
- Coler R. Therapeutic immunization against Mycobacterium tuberculosis is an effective adjunct to antibiotic treatment. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Picas, Jordi (Archivel Farma, Badalona, Spain). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 5.
- Vilaplana C, Gil O, Caceres N, et al. Prophylactic effect of a therapeutic vaccine against TB based on fragments of Mycobacterium tuberculosis. PLoS One. 2011;6(5):e20404. doi: 10.1371/journal.pone.0020404.
- Vilaplana, Cris (Experimental Tuberculosis Unit, Barcelona, Spain). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 20.
- von Reyn, Ford (Geisel School of Medicine, Dartmouth University, Hanover, NH). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 25.
- von Reyn F, Mtei L, Arbeit R, et al. Prevention of tuberculosis in bacille Calmette-Guérin-primed, HIV-infected adults boosted with an inactivated whole-cell mycobacterial vaccine. AIDS. 2010;24(5):675–685. doi: 10.1097/QAD.0b013e3283350f1b.
- von Reyn F. Polyantigenic DAR-901: an inactivated whole cell vaccine for the prevention of tuberculosis. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Evans, Tom. (Aeras, Rockville, MD). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 7.
- de Bruyn G, Garner P. Mycobacterium vaccae immunotherapy for treating tuberculosis. Cochrane Database Syst Rev. 2003;(1):CD001166. Review. doi:10.1002/14651858.CD001166.
- Katoch K, Singh P, Adhikari T, et al. Potential of Mw as a prophylactic vaccine against pulmonary tuberculosis. Vaccine. 2008;26(9):1228–34. doi: 10.1016/j.vaccine.2007.12.025.
- Bhaskar, Sangeeta (National Institute of Immunology, Department of Biotechnology, New Delhi, India). E-mail with: Mike Frick (Treatment Action Group, New York, NY). 2013 March 30.
- Bhaskar, Sangeeta (National Institute of Immunology, Department of Biotechnology, New Delhi, India). Personal communication with: Mike Frick (Treatment Action Group, New York, NY). 2013 April 1.
- Knight G. Global cost effectiveness of new tuberculosis vaccines: a modeling study. Paper presented at: TB vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Kaufmann S. Fact and fiction in tuberculosis vaccine research: 10 years later. Lancet. 2011;11(8):633–40. doi: 10.1016/S1473-3099(11)70146-3.
- Hanekom W. Correlates of risk of TB disease in adolescents. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Zak Dan. Systems analysis of TB vaccines and TB disease risk. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Minassian A, Ronan E, Poyntz H, et al. Preclinical development of an in vivo BCG challenge model for testing candidate TB vaccine efficacy. PLoS One. 2011;6(5):e19840. doi:10.1371/journal.pone.0019840.
- McShane H. Human mycobacterial challenge models. Paper presented at: TB Vaccines Third Global Forum; 2013 March 25–27; Cape Town, South Africa.
- Barker L, Hessel L, Walker B. Rational approach to selection and clinical development of TB vaccine candidates. Tuberculosis. 2012;92(S1):S25–29. doi: 10.1016/S1472-9792(12)70009-4.
- Jiménez-Levi E. 2012 report on tuberculosis research funding trends, 2005–2011. New York: Treatment Action Group; 2012. Available from:
http://www.treatmentactiongroup.org/tbrd2012. (Accessed April 10, 2013).
[i] In India, category I refers to drug-susceptible TB patients receiving standard first-line anti-TB drugs (isoniazid, rifampicin, ethambutol, pyrazinamide). Category II refers to patients who did not complete category I treatment but are not confirmed DR-TB cases. Under category II, patients receive a regimen that includes first-line anti-TB drugs with the addition of streptomycin.

